AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Hybridization of atomic orbitals4/19/2023 
The teaching of chemical bonding is a unique opportunity in the science curriculum to achieve these goals, but this approach requires an awareness of the logical and historical underpinnings of the different models. This approach encourages higher order thinking, deepens understanding and engages students with a more open, dynamic portrayal of science as the vital human endeavour it is. Instead of classifying different models as simplistically right or wrong, we need to help our students compare alternative scientific perspectives in terms of strengths and weaknesses. One of the most important goals in science education should be to demonstrate that science is not the static, black and white body of knowledge found in the average textbook. The best reason to teach hybrid orbitals, as a component of Linus Pauling’s VB theory, is to compare and contrast it with Lewis structures and the delocalized, molecular orbital understanding of bonding. This exercise, if done correctly, teaches students a valuable lesson about the nature of scientific models. 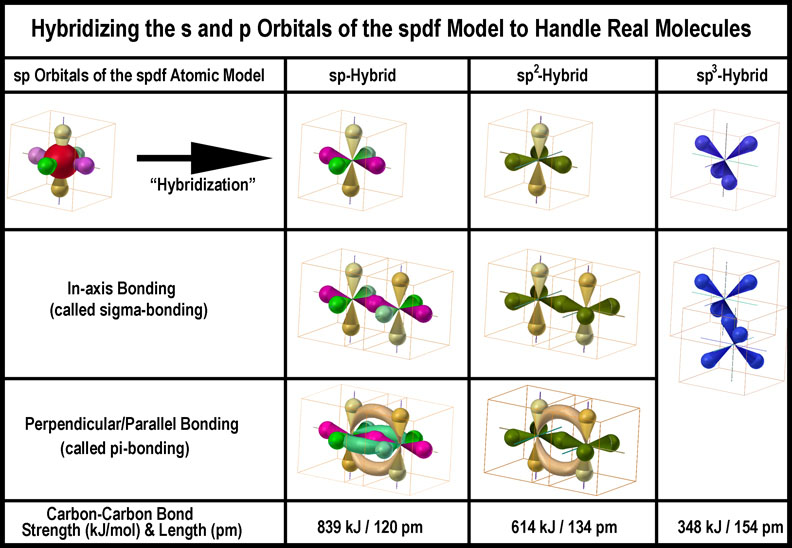
This point is well made by Nivaldo Tro:Ĭhemical bonding is probably the only place in the curriculum where we teach multiple models for a single phenomenon. 
Just getting students through with a fairy tale understanding of chemical bonding robs us of an opportunity to expose them to some profound lessons in the nature and development of the modern science of chemistry. As a high school teacher, I do not feel qualified to contribute to this debate among professional chemists, but I would like to argue that making our students aware that such debates exist in contemporary science is essential in shaping a deeper and more authentic view of the nature of science in our students.ĭespite their shortcomings, hybrid orbitals should not be taught as merely a flawed and simplified story we tailor to the limited powers of the teenaged mind. It is clear from this discussion that the value of localised models of chemical bonding is still an open question. In contrast, Landis, Weinhold and Tro contend that a hybrid orbital approach, as a component of valence-bond (VB) theory, is still properly used by contemporary chemists to model electron densities and bonding, and that introducing these concepts does not get in the way of the future learning of students, but provides a secure foundation for more advanced work. 5 Grushow argues that the concept of hybrid orbitals is outdated, with very few valid applications in contemporary chemistry, and only ingrains an incorrect understanding of bonding which students will have to overcome in future learning. The value of teaching hybrid orbitals has been the topic of recent articles in the Journal of Chemical Education 2,3,4 and in this publication. 
Nevertheless, combining every theoretical argument available, however shaky, with experiments of many kinds, chemists have built up one of the great intellectual domains of mankind and have acquired great power over nature, for good or ill. Our concepts are often ill-defined, our rules and principles full of exceptions, and our reasoning perilously near being circular. Chemistry has a method of making progress which is uniquely its own and which is not understood or appreciated by non-chemists.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
